What is Real World Data?
Characteristics and How to Use it
“Real World Data (RWD)” are now attracting attention in order to allocate medical costs that are increasing along with the aging society.
RWD are the collection and analysis of data obtained from actual clinical settings.
Utilization of real world data by medical institutions and municipalities can help improve the quality of medical care and restore the soundness of hospital management.
In this article, we will explain what RWD are and how we can use them.
In order to provide better medical care, we would like to suggest you consider using RWD in the fields of new drug development, research, and hospital management.
What is RWD?
What kind of information are considered as RWD in the first place?
First, we would like to start with introducing the overview of RWD.
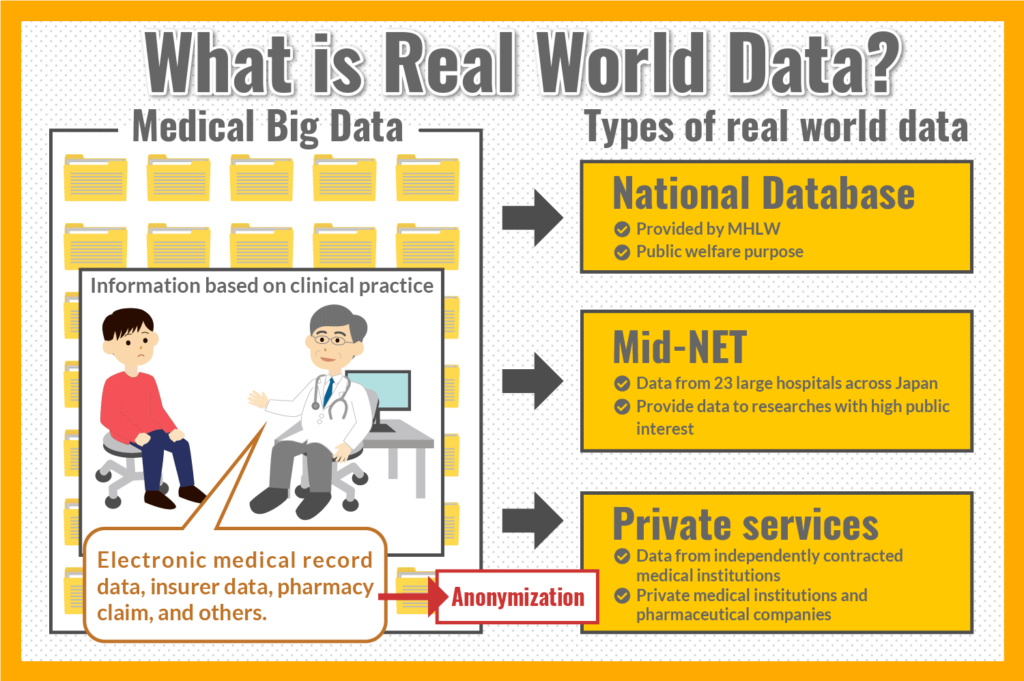
RWD refer to Medical Big Data
RWD refer to medical big data, which are collections of information based on medical treatments obtained at clinical settings, such as pharmacy claim data, insurer data, electronic medical record data, and so on.
Patients with multiple diseases and/or undiagnosed symptoms are all recorded and primarily it is created for purposes other than research.
Although detailed symptoms and prescription histories are recorded, patient names are anonymized so that more detailed medical data can be obtained while protecting privacy.
However, it is important to note that the data is not created for research purposes and is different from the data collected using methods that ensure reliability.
It includes incomplete and biased data, hence it is not suitable to be used in research for medical statistical purposes.
Different Types of RWD
Currently, multiple institutions are handling RWD.
Following services are typical examples.
- NDB (National Database)
RWD for public interest purposes, provided by the Ministry of Health, Labour and Welfare (MHLW).
Data are provided only to government agencies, universities, independent administrative agencies that conduct research, public interest corporations under the jurisdiction of the government, national government agencies, etc. - MID-NET
RWD collected from electronic medical records and other data from 23 large hospitals across Japan, used for post marketing surveillance or research of high public interest.
Data are provided only to surveys of the actual status of drugs which MHLW has requested development, and to research funded by public research funds from the national government, municipalities, and independent administrative agencies.
As explained above, it is necessary to clear a very big hurdle to receive RWD information operated by public institutions.
Furthermore, in order to receive data, it was required to prepare a dedicated room with security, a server to handle huge amount of data, and an expensive analysis environment.
Therefore, it was considered difficult for general medical institutions to obtain RWD.
However, in recent years, in addition to those public institutions, some services utilizing RWD have been seen in the private sector.
Private companies are building RWD by collaborating with independently contracted medical institutions and municipalities and receiving their medical data and other information.
Non-personal information is extracted from the submitted information with anonymizing technology, allowing companies to accumulate data while protecting privacy.
With these private services, medical institutes and pharmaceutical companies that have not been able to use RWD in the past can now receive information.
Analysis reports can be created if necessary, so it can lead to further improvement of medical care quality and the restoration of the soundness of hospital management.
The Purposes of RWD
RWD are highly effective within medical industry, but for what purpose are they being used in the first place?
In this section, we will explain the purpose of secondary utilization of medical data.
1. Improving Efficiency of New Drug Development within Pharmaceutical Companies
RWD are the most useful in the development area of pharmaceutical companies.
In the pharmaceutical research phase, RWD are used to analyze risk factors by referring to a database of entry themes.
Furthermore, at the development step, RWD can be useful to increase evidence, streamline the development process, and formulate marketing methods.
Also, RWD can bring great benefits to the clinical trial step. Traditional clinical trial methods required both “test drug group” and “control group” patients.
Controlled trial, which compares validity and safety by examining the both results, is not an efficient method since it is time consuming and costly.
On the other hand, it is possible to substitute control group for data if we can use RWD.
Since only the patients in “test drug group” need to be recruited for clinical trials, the time and cost required of clinical trials would be significantly reduced.
In this method, it enables efficient development of new drugs even in cases where the number of patients is limited, such as for the intractable or rare diseases.
2. Speeding up Post Marketing Surveillance on Drug Research
RWD are used not only at a new drug development step but also for post marketing surveillance.
If you use RWD that include many background factors, it is possible to collect information that cannot be conveyed by sales data alone and improve the drug usage insight.
It conveys the number of patients, laboratory values, complications, and other factors that can help you discover your company’s issues and create future marketing strategies.
Conducting these post marketing surveys on your own can be very costly and time consuming; however, by utilizing RWD, you will be able to conduct research and analysis quickly and at low cost.
3. Medical Care Quality Improvement and Soundness of Hospital Management
Not only pharmaceutical companies are the ones who can receive benefits from RWD.
RWD have a lot of benefits for medical institutions as well.
By using RWD, it becomes possible to perform various analyses, such as visualizing patients with polypharmacy in a hospital and quantifying the effects of medical treatment.
It will be useful in multiple areas, including measures to improve the quality of medical care, optimize medical fees, and devise measures to improve hospital management policies.
As previously mentioned, it is important for medical institutions not only to provide RWD but also to use the data in order to provide better medical care.
4. Measures Against Rare Diseases and Intractable Diseases
Rare and/or intractable diseases are known for their sometime very small number of patients with the mechanisms of disease onset not fully known yet in many instances.
Therefore, even if a patient visits a medical institution, there are many cases that the patient cannot receive adequate treatment because the development of drugs is delayed or the disease is not appropriately recognized.
By providing and sharing information about these intractable diseases to RWD, it should help medical institutions and municipalities to improve the accuracy of their data on patients with intractable diseases.
As RWD become more widespread, it will be possible to provide appropriate treatment to patients with rare and/or intractable diseases who have been overlooked until now.
Differences Between Clinical Trials and RWD
RWD are not the only database collecting patient information.
Clinical trial data are also created by collecting actual patient cases.
However, it is important to distinguish between clinical trial data and RWD because they are completely different. Now, let’s take a look at the differences between clinical trials and RWD, which can be a bit confusing.
Characteristics of Clinical Trial
In the first place, it is important to understand that clinical trial data is “experimentally collected data” based on an implementation plan.
Only the data from trials conducted on “patients/populations with a case that meets the criteria” are collected, not on an unspecified population.
The main characteristic is that information is collected for the purpose of verifying specific efficacy and safety, and data are collected in accordance with GCP, the rules that should be followed as clinical trials are conducted, and various guidelines.
Characteristics of RWD
In contrast, it is a major characteristic of RWD that the data are collected in daily medical practices.
Followings are the types of data which are collected as RWD.
- Electronic medical records
- DPC data
- Health claims
- Patient registration/ registry
- Patient-generated health data, such as wearable devices and home health equipment
etc.
Examples of RWD Utilization
Let’s look at examples of RWD utilization:
1. Analyzing the Actual Drug Prescription Status
A pharmaceutical company A wants to know the actual prescription status of their drugs.
Although the company knew the total number of drug sold, they did not have any data on each patient or on concomitant drugs, so RWD were used to find out the actual prescription status.
As a result, they were able to analyze both new and continuing patients with minimal cost and time, and evaluate information about switching/discontinuation of prescriptions.
They also found out the number of patients and the amount of prescriptions for each indication, and the analyzed data are used to identify future issues for the company.
2. Understanding the Size of the Market before Developing New Drugs
A pharmaceutical company B, intending to develop a new drug.
RWD were used to find out about diseases, medication trends, and the number of treated patients in their entry area.
Previously, they could not track medications and procedures per patient; however, RWD will convey not only the number of patients and procedures but also the gender and age distribution.
It was very useful in providing more detailed references to consider entering a new field.
RWD have a Big Impact on the Pharmaceutical and Treatment Fields
RWD are medical big data which collect information based on medical practices in clinical settings.
It is useful not only in the pharmaceutical field, where drug prescription status is tracked and clinical trials are conducted, but also in improving the services and operations of medical institutions. It will be used more and more in the future.
We, Medical Data Vision Co., Ltd., offer multiple services using RWD.
If there is anything we can help you with utilizing data, such as understanding the actual status of prescriptions or collecting various indicators related to risk assessment, please feel free to contact us.
For More Information, Please Contact Us Here
About Japanese Healthcare System

What you need to know about the healthcare system in Japan before using the data.
SERVICE

In addition to various web tools that allow you to easily conduct surveys via a browser using our medical database, we offer data provision services categorized into four types to meet your needs and challenges: "Analysis reports" "Datasets," "All Therapeutic Areas Data Provision Service," and "Specific Therapeutic Areas Data Provision Service.

© Medical Data Vision Co., Ltd. All Rights Reserved.





