What is a Clinical Trial? And what are the Virtual Clinical Trials that have been attracting attention in recent years
In this article, we intend to explain the process of creating a new drug and the flow of the studies and trials, as well as what is the difference between clinical and non-clinical studies. You may also have heard of “virtual clinical trials” but do not know what it really means and implies. This article also provides a detailed overview of “virtual clinical trials” and their benefits.
What is a clinical trial?

Clinical trials are clinical studies conducted in hospitals primarily to obtain approval from the government for a new drug. The drug is tested on patients or healthy adults to confirm its effectiveness and safety.
Pharmaceutical companies submit the results of the clinical trial to the Ministry of Health, Labour and Welfare (MHLW). Only after they have been reviewed and approved by the government, do new drugs become available for use by the public.
Only medical institutions that meet the requirements specified in Article 35 of the “Good Clinical Practice (GCP)” are allowed to conduct clinical trials. In addition, the responsible person for the clinical trial must be a physician who meets the requirements specified in Article 42.
What is a clinical study?
A clinical study is a study conducted to confirm the efficacy and safety of a new drug, medical device, or treatment. Among clinical studies, a clinical trial is a study conducted on “human subjects” at a medical institution to obtain approval from the government for a new drug.
To research and develop a new drug and bring it to market, non-clinical and clinical studies must be conducted under the “GCP”.
Non-clinical studies
Non-clinical studies are studies conducted on animals.
Medicines used in therapeutics are made from plant-derived ingredients, ingredients found in soil fungi and microorganisms, as well as chemically synthesized chemicals and substances purified by biotechnology. Under the assumption, that these ingredients and substances are beneficial to humans, studies are conducted on “animals” to determine their efficacy, safety, toxicity, and feasibility.
The animals tested include mice, rats, guinea pigs, rabbits, dogs, and pigs.
Clinical studies
A clinical study is a study conducted on “human subjects” after a non-clinical study has been performed, either on animals or “in-vitro”, that is to say in laboratory test setting.
In clinical studies, drug candidates with potential efficacy and effectiveness in “human subjects” are selected based on data on pharmacological effects, pharmacokinetics, adverse events, etc. obtained in non-clinical studies. Then, the drug candidates are used on “human subjects” to confirm efficacy, safety, dosage, and administration.
Clinical trials are conducted in three phases: Phase I, Phase II, and Phase III.
How a new drug is produced
A new drug is completed in the following five steps.
- Basic study
- Non-clinical study
- Clinical study
- Review and approval
- Post Marketing Surveillance
We will explain this in detail below.
1. Basic study
In basic studies, the first step in creating a new drug, drug candidates from plant-derived ingredients, soil bacteria, microorganisms, sea organisms, etc. are searched for, and their structures, properties, and states are investigated.
Basic studies take about two to three years to complete.
2. Non-clinical study
Once a study on ingredients and substances is completed, non-clinical studies on “animals” are conducted. In non-clinical studies, the following studies are performed.
- Pharmacology & efficacy study
This study is done to understand what doses are effective, how effective those different doses are, and how to utilize the drug. - Pharmacokinetic study
This study examines how drugs are absorbed into the body, distributed throughout the tissues through the bloodstream, and metabolized and eliminated by the liver. Drug interactions are also studied. - General pharmacological study
This investigates what parts of the body the drug acts on and how it acts on them. Safety outside of the scope of pharmacological effects will also be investigated. - General and special toxicity study
This is to examine the safety and toxicity of the product over short, medium, and long dosing periods. Carcinogenicity and its effects on fetal development will also be examined.
Non-clinical studies take about three to five years to confirm efficacy, safety, and toxicity.
3. Clinical Study
Once the feasibility is confirmed in non-clinical animal studies, clinical studies are conducted on human subjects. Clinical studies are conducted in the following three phases.
- Phase I clinical trial (clinical pharmacology study)
Testing a drug in a small number of healthy adults to determine safety and pharmacokinetics. - Phase II clinical trial (exploratory study)
Testing a drug in a small number of patients to study its efficacy and safety. - Phase III clinical trial (validation study)
Testing a drug in a large number of patients to study its efficacy and safety.
Clinical trials are conducted on healthy adults and patients over a period of three to seven years to confirm the efficacy, safety, dosage, and administration of the drug.
4. Review and approval
Once clinical trials are completed and efficacy and safety are confirmed, an application for approval is submitted to the MHLW. “The Pharmaceutical Affairs and Food Sanitation Council”, an advisory body to the MHLW, deliberates on the application, and it takes one to two years for approval. Only after the approval is granted is the drug considered to be a pharmaceutical product.
5. Post Marketing Surveillance
After a drug is put on the market and widely used as a pharmaceutical product, the effectiveness and safety of the drug continue to be investigated. On the market, the drug can be used by many more patients than in clinical trials. Data on efficacy and safety obtained from the use of the drug on a large number of patients are fed back to the pharmaceutical company and utilized for the improvement and development of the drug.
Clinical trial flow
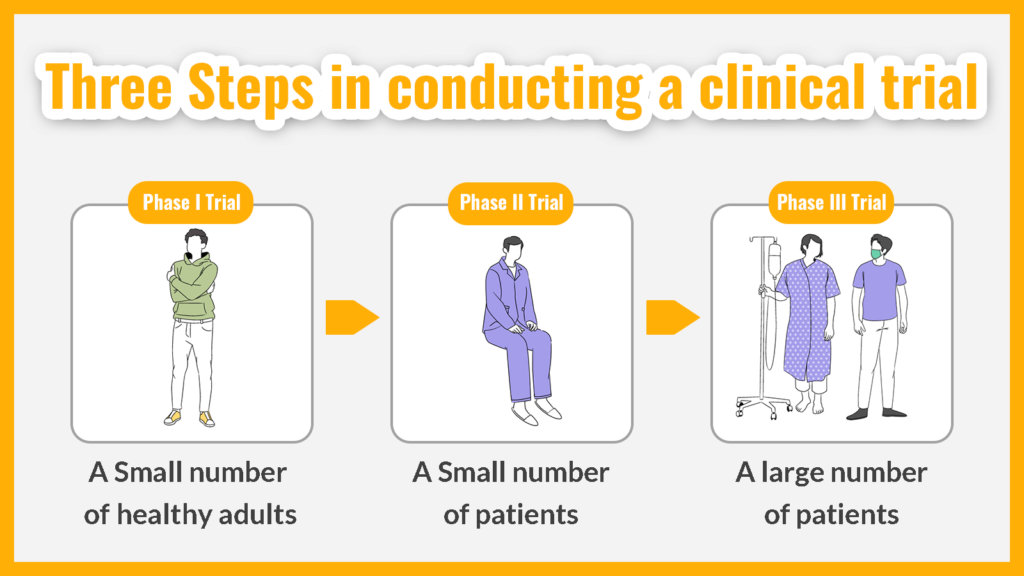
Clinical trials are conducted in three steps, “Phase I trial”, “Phase II trial”, and “Phase III trial”, and take three to seven years to complete. Below is a description of the subjects and study content of each trial.
1. Phase I trial
In Phase I studies, an investigational drug is used in a small number of healthy adults to determine its safety and pharmacokinetics. The drug is administered in very small doses, starting at doses much smaller than those expected to be used, and gradually increasing the dose.
The reason for using healthy adults is that it is impossible to determine whether side effects if they occur, are caused by the investigational drug or by a combination of the drug and the subject’s original medication. To determine the safety and pharmacokinetics of the investigational drug under the same conditions as similar conditions as possible, the drug is first checked in healthy adults.
2. Phase II trial
In Phase II trials, an investigational drug is used in a small number of patients to determine how well it works. How much and when to administer or take the drug is determined to be the most effective and cause the fewest side effects.
When examining the efficacy and dosing methods, different doses are compared. Phase II trials are also called “exploratory clinical trials”.
3. Phase III trials
In Phase III trials, the investigational drug is used in a large number of patients based on data from Phase I and Phase II trials to determine how well it works. In addition to comparing the investigational drug with a controlled drug (the standard drug currently used in the treatment), placebos (fake drugs) that do not contain the active ingredient are used to examine efficacy so that preconceptions and assumptions will not change the results.
The final safety of the product is also confirmed by using it on a larger number of patients.
Virtual clinical trials in the spotlight
In recent years, virtual clinical trials have been attracting attention.
The outbreak of Coronavirus disease (COVID-19) has led to online medical treatment and drug prescriptions in the medical field. The wave of digitization is not only taking place in the medical field but is also taking place in the field of drug development.
Clinical trials are generally conducted at medical institutions, so subjects have to go to medical institutions and may need to be hospitalized. Because hospital visits and hospitalization are burdensome for subjects, it was sometimes difficult to find subjects and the clinical trial did not progress.
To solve the problem of “clinical trials not progressing due to lack of subjects”, the MHLW is developing regulations to enable clinical trials that do not depend on subjects visiting hospitals, which is called Decentralized Clinical Trial (DCT) to be conducted in Japan.
What is a virtual clinical trial?
A virtual clinical trial is a clinical trial that can be conducted at home without the subject having to come to the hospital. Virtual clinical trials are conducted using the following systems and tools.
- eConsent
It is a system to obtain via electronic methods the consent by the patient and/or the patient’s family to participate in the clinical trial, after reviewing the detailed explanations regarding the clinical trial methods and purposes. - Telemedicine
A medical practice in which a doctor’s consultation, drug prescription, etc. can be received online using video calls through electronic devices such as PCs and smartphones. - Wearable technology
A computer device is worn on the arm, leg, head, etc. to obtain biometric and exercise information. - electronic Patient Reported Outcome (ePRO)
A system that allows healthcare providers to evaluate in real-time the health status recorded by patients via smartphones, tablets, etc.
Advantages of virtual clinical trials
Because virtual clinical trials do not require hospital visits, they are easier to recruit for and as well as conduct than general clinical trials. Additionally, the withdrawal rate will be lower because subjects will not be obliged to go to the hospital or stay in the hospital. As a result, it is expected to shorten the time it takes to develop a new drug.
Since subjects continue their usual lifestyle during the clinical trial, realistic data can be collected from their daily lives, rather than data from the unusual space of a medical institution. By utilizing wearable technology and ePRO, data on subjects’ health and physical activity in their daily lives can also be collected.
Through virtual clinical trials, we can expect to develop drugs that reflect the daily lives of patients in the future.
Summary
New drugs take nine to seventeen years from development to approval, and clinical trials alone take three to seven years. Clinical trials are generally conducted at medical institutions, so subjects need to go to the hospital or be hospitalized, which makes it difficult to conduct them.
In recent years, however, attention has focused on virtual clinical trials, in which subjects can undergo clinical trials at home without having to come to the hospital. The advantage of virtual clinical trials is that it is easier to recruit subjects and shorten the development period.
The MHLW is working to develop regulations that will allow distributed clinical trials (DCT) to be conducted in Japan without requiring subjects to visit hospitals, so we should keep a close eye on this trend.
Medical Data Vision Co., Ltd. provides services such as tools for analyzing the number of patients and the volume of prescriptions in medical databases and analysis tools specialized for the oncology field. Our medical data is also used for clinical trials and clinical research. Please contact us for more information.
For More Information, Please Contact Us Here
About Japanese Healthcare System

What you need to know about the healthcare system in Japan before using the data.
SERVICE

In addition to various web tools that allow you to easily conduct surveys via a browser using our medical database, we offer data provision services categorized into four types to meet your needs and challenges: "Analysis reports" "Datasets," "All Therapeutic Areas Data Provision Service," and "Specific Therapeutic Areas Data Provision Service.

© Medical Data Vision Co., Ltd. All Rights Reserved.





