
Trends in the Number of Patients and Duration of Regimen Use in First-Line Treatment for Colorectal Cancer
March each year marks “Colorectal Cancer Awareness Month,” established in the United States in February 2000, during which the international Blue Ribbon campaign is promoted. In Japan, several organizations, including the Japan Cancer Society, conduct awareness activities, such as illuminating landmarks in blue as part of the Blue Ribbon Light-Up initiative.
Colorectal cancer consistently ranks among the most common cancers in Japan in terms of incidence. While surgical resection remains the foundation of treatment, therapeutic approaches have become increasingly sophisticated with the advancement of individualized pharmacotherapy based on genetic alterations and patient characteristics. In recent years, optimization of perioperative management has become an important area of research. For Stage III colon cancer, postoperative adjuvant chemotherapy is the standard of care. Meanwhile, in locally advanced or high-risk cases, the utility of preoperative chemotherapy aimed at controlling micrometastases and achieving downstaging has been demonstrated in clinical trials, and its selective introduction is under consideration.
Using MDV data, we analyzed trends from 2023 to 2025 in the number of patients receiving major first-line regimens and the duration of regimen use, stratified by the presence or absence of perioperative pharmacotherapy (NAC/ADJ) and by sex.
Analysis Conditions
- Target population: Patients with Stage III or higher disease in whom initiation of second-line treatment was confirmed
- Observation period: 2023–2025
- Total number of patients: 31,133 (cumulative over the study period)
1. Major Regimens (With NAC/ADJ, Male)
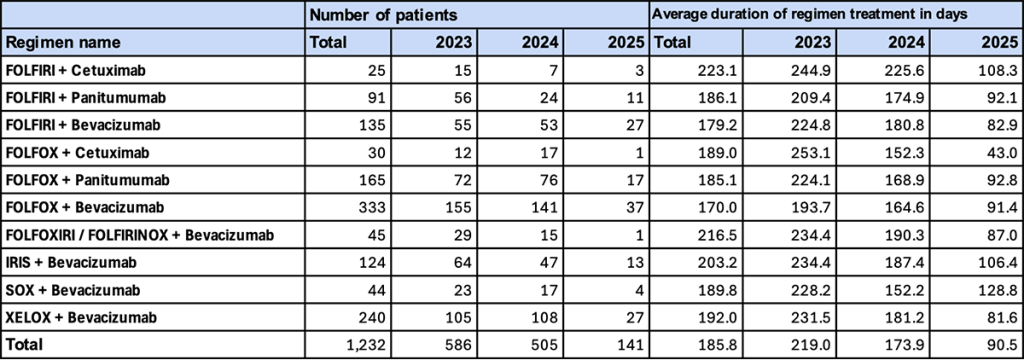
- Trend in Number of Patients
“FOLFOX + bevacizumab” had the highest number of patients throughout the entire period. In 2025, patient numbers decreased across all regimens, partly due to the data aggregation period; however, there was no change in the relative ranking among regimens. - Duration of Regimen Use
As of 2023, “FOLFOX + cetuximab” (253.1 days) and “FOLFIRI + cetuximab” (244.9 days) showed relatively long treatment durations. By 2024, an overall shortening trend was observed.
2. Major Regimens (With NAC/ADJ, Female)
This section presents trends in first-line treatment among female patients who received perioperative pharmacotherapy (NAC/ADJ).
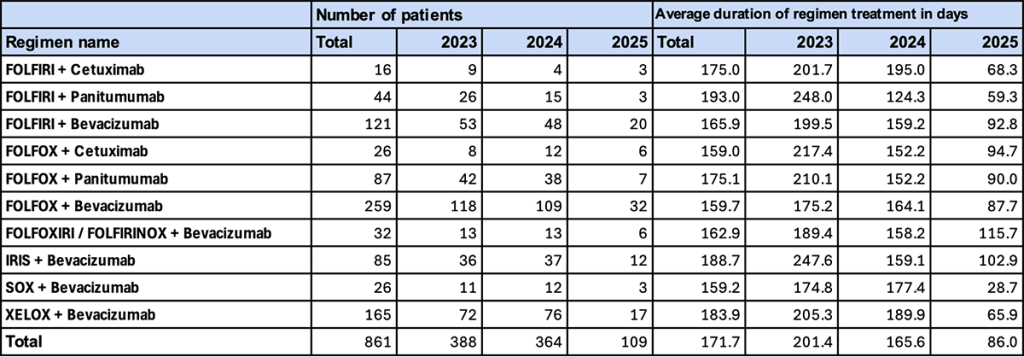
- Trend in Number of Patients
Similar to males, “FOLFOX + bevacizumab” (118 patients in 2023; 109 in 2024) had the highest number of patients, followed by “FOLFIRI + bevacizumab” (53 in 2023; 48 in 2024). - Duration of Regimen Use
In 2023, “FOLFIRI + panitumumab” (248.0 days) and “IRIS + bevacizumab” (247.6 days) showed relatively long durations. In 2024, most regimens converged to approximately 150–180 days. In 2025, durations shortened further, with several regimens falling below 100 days.
3. Major Regimens (Without NAC/ADJ, Male)
This section presents trends among male patients who did not receive perioperative pharmacotherapy (NAC/ADJ) and who underwent first-line treatment at initial diagnosis or recurrence.
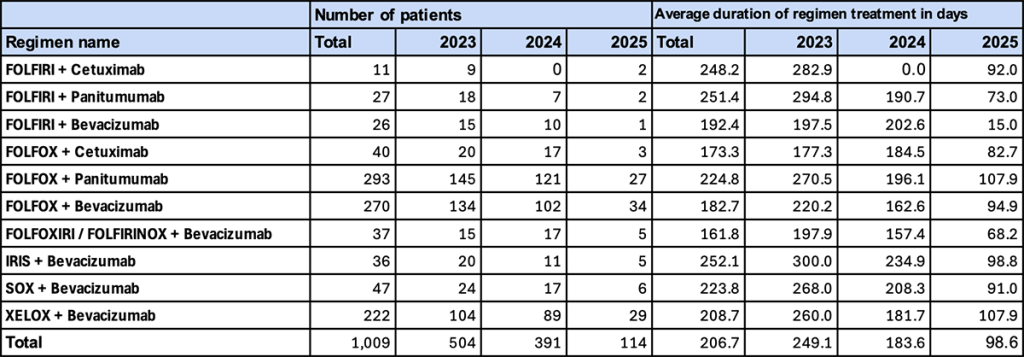
- Trend in Number of Patients
“FOLFOX + panitumumab” was the most frequently used regimen, followed by “FOLFOX + bevacizumab.” - Duration of Regimen Use
In 2023, “IRIS + bevacizumab” (300.0 days) and “FOLFIRI + panitumumab” (294.8 days) showed notably long treatment durations. From 2024 onward, a marked shortening was observed.
4. Major Regimens (Without NAC/ADJ, Female)
This section presents trends among female patients who did not receive perioperative pharmacotherapy (NAC/ADJ) and who underwent first-line treatment at initial diagnosis or recurrence.
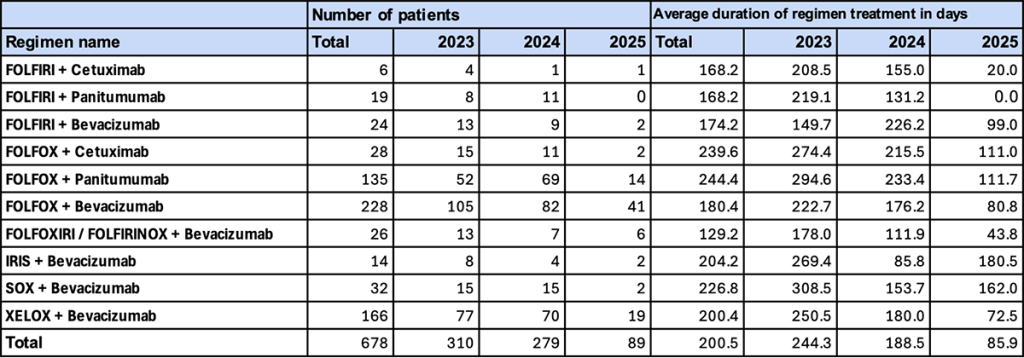
- Trend in Number of Patients
“FOLFOX + bevacizumab” was the most frequently used regimen (105 patients in 2023; 82 in 2024), followed by “XELOX + bevacizumab” (77 in 2023; 70 in 2024). In contrast to males, “FOLFOX + panitumumab,” which was common among males, ranked third. - Duration of Regimen Use
In 2023, “SOX + bevacizumab” (308.5 days) showed a particularly long duration. In 2024, durations decreased to approximately 150–230 days. In 2025, with the exception of “IRIS + bevacizumab” (180.5 days), many regimens shortened to approximately 80–110 days.
Overall Interpretation of the Graphs
Across all graphs, a common trend is the shortening of average regimen duration from 2023 to 2024. In 2025, durations may have been calculated as even shorter due to the partial observation period. However, the recent shortening trend may reflect the increasing sophistication of treatment management, including earlier assessment of treatment response, optimization of strategies incorporating molecular targeted therapies, and earlier transition to subsequent lines of therapy based on disease progression or resectability assessment.
Differences in regimen selection were observed depending on treatment background. Among patients who received perioperative pharmacotherapy (NAC/ADJ), “FOLFOX + bevacizumab” was the most frequently used regimen in both males and females, suggesting a tendency toward anti-VEGF combination therapy in consideration of recurrence control and disease management.
In contrast, among males without a history of perioperative pharmacotherapy (NAC/ADJ), “FOLFOX + panitumumab” was most frequently used, suggesting that anti-EGFR antibody combination therapy may be selected in consideration of tumor shrinkage and response rate.
Among females without a history of perioperative pharmacotherapy (NAC/ADJ), “FOLFOX + bevacizumab” was again the most frequently used regimen, indicating that treatment selection in real-world clinical practice may reflect differences in patient background and treatment context.
References
- Japan Society for Cancer of the Colon and Rectum. Japanese Society for Cancer of the Colon and Rectum (JSCCR) Guidelines 2024 for the Treatment of Colorectal Cancer (for Physicians).
- Japanese Society of Medical Oncology. Clinical Practice Guidelines for Cancer Treatment: Process for Determining First-Line Treatment Strategy.
http://www.jsco-cpg.jp/colorectal-cancer/algo/#V-2
Note: This article was published on March 5, 2026.
Data survey and analysis tailored to your specific requests
Databases, data analysis requests, and more.
© Medical Data Vision Co., Ltd. All Rights Reserved.





