A collection of videos including seminars, interviews, and product introductions related to RWD.
-

Building a Strategic Marketing Plan in Pharma | The Real-World Talk
A strong marketing plan is more than a beautiful slide deck, it is a strategic story grounded in data, built to drive real behavior change. In this episode of The Real-World Talk, Jeffrey Schnack ( President of 3Rock KK) draws on 25 years of experience working with pharmaceutical and medical device brand planning teams in Japan to share what truly separates an effective brand plan from a forgettable one. He explores why logical flow, robust data foundations and cross-functional communication are the three pillars of any credible plan, how real-world data can reveal the evolving behaviors of prescribers and patients across a 3-to-5 year horizon, and why the healthcare crossroads, where patient experience meets customer experience, must sit at the heart of every actionable strategy. The conversation also addresses the growing role of AI in marketing planning. While AI can significantly accelerate plan creation and structure, Jeffrey emphasizes that the real value lies in critically challenging assumptions, validating strategies in the field, and refining the final plan to drive real impact. A practical and forward-looking conversation for any pharma marketing or market access team looking to build plans that are not just approved but actually implemented. Meet the Expert Jeff began his career with a boutique consulting firm focusing on new market business development. After launching the JPAC subsidiary of a German industrial manufacturer, he led the first overseas subsidiary of the East Japan Railways Group, creating a US consumer business that won a #2 ranking in the Nikkei Shimbun New Product of the Year competition. He now focuses on strategy development, implementation and capability-building for clients in the pharma and healthcare industry. Jeff has taught courses on Brand and Project Management in the CUMOT program at the Tokyo University of Technology, and Technology Marketing at the QREC Entrepreneurship Center of Kyushu University. Recently he has spoken overseas at digital healthcare events "Medicine X" at Stanford University and "Doctors 2.0 & You" in Paris. He also contributes to industry press such as Monthly Mix. Contact For further information about MDV’s real-world data solutions, or to discuss potential collaboration opportunities, please contact: Medical Data Vision Co. Ltd (MDV) Overseas Business Development Office Mail to:rwd_customer@mdv.co.jp Disclaimer The views and opinions expressed by the speakers in this video are the individual views and opinions of their respective speakers and are not a reflection of the official policy or position on any topic of our company or any other company (including any company with which the speaker may be employed or affiliated). The statements made by the speakers in this interview should be regarded as matters of opinion and not as representations of fact. We are unable to guarantee with certainty the veracity of any of the statements made by the speakers in this video. This video should be regarded as an honest review of our company’s products and services from the perspective of a real user of our company’s products and services. This video was prepared for general informational purposes only and our company assumes no liability for any errors or omissions in the content, or for any actions taken based on the information provided. The speakers in this video received no compensation, reward or any other form of inducement from our company to participate in this interview or to endorse any of our products or services. This video and any content contained within this video is protected by copyright law. Unauthorized use or distribution is strictly prohibited.
2026.04.28 10:27
-

EHR Data in Japan : Unlocking Real-World Insights | The Real-World Talk
What does it really take to leverage real-world data (RWD) in Japan? In this episode of The Real-World Talk, Dr. Keiko Asao (Kappa Medical) and Dr. Yuki Koyama (TXP Medical) share their unique journeys as physicians and data experts across Japan, the US, and Australia, and explain how these experiences shaped their perspective on real-world evidence. They explain how TXP provides high-quality Japanese EHR datasets, enabling teams to analyze patient journeys, uncover unmet needs, and make informed strategic decisions. The conversation also explores the future of RWD in Japan and how connected data and advanced analytics can transform healthcare insights. Contact For further information about MDV’s real-world data solutions, or to discuss potential collaboration opportunities, please contact: Medical Data Vision Co. Ltd (MDV) Overseas Business Development Office Mail to:rwd_sales@mdv.co.jp Disclaimer The views and opinions expressed by the speakers in this video are the individual views and opinions of their respective speakers and are not a reflection of the official policy or position on any topic of our company or any other company (including any company with which the speaker may be employed or affiliated). The statements made by the speakers in this interview should be regarded as matters of opinion and not as representations of fact. We are unable to guarantee with certainty the veracity of any of the statements made by the speakers in this video. This video should be regarded as an honest review of our company’s products and services from the perspective of a real user of our company’s products and services. This video was prepared for general informational purposes only and our company assumes no liability for any errors or omissions in the content, or for any actions taken based on the information provided. The speakers in this video received no compensation, reward or any other form of inducement from our company to participate in this interview or to endorse any of our products or services. This video and any content contained within this video is protected by copyright law. Unauthorized use or distribution is strictly prohibited. EHR data video minutes and chapters 00:00 03:48 What international experience brings you in perspective to RWD? 08:35 What are the main challenges for RWD in Japan, and how do you see this evolving? 13:51 With the increase use of EHR data, what new insights or use cases are now becoming possible? 18:33 Looking ahead, where do you see the most promising opportunities for RWD in Japan? 24:30 Can you share examples of how EHR data is being effectively used in real-world settings? 27:30 What are some best practices to keep in mind when working with EHR data for evidence generation? 30:44 Finally, what key trends or innovations do you expect to shape the future of EHR-based research?
2026.03.31 33:15
-

Designing Strong RWE Research Questions | The Real-World Talk
Real-world data only becomes meaningful when it is guided by the right question. In this episode of The Real-World Talk, Dr. Keiko Asao (Kappa Medical K.K.) shares her perspective on what truly defines a strong and scientifically valuable research question when working with real-world evidence in Japan. She discusses why relevance, feasibility and clinical grounding are essential, how understanding the characteristics and limitations of Japanese datasets shapes what is realistically achievable, and why strategic thinking and transparency are critical throughout the study process. This conversation offers practical and experience-based guidance for teams seeking to design robust, credible and decision-relevant RWD studies in the Japanese context. Contact For further information about MDV’s real-world data solutions, or to discuss potential collaboration opportunities, please contact: Medical Data Vision Co. Ltd (MDV) Overseas Business Development Office Mail to:rwd_sales@mdv.co.jp Disclaimer The views and opinions expressed by the speakers in this video are the individual views and opinions of their respective speakers and are not a reflection of the official policy or position on any topic of our company or any other company (including any company with which the speaker may be employed or affiliated). The statements made by the speakers in this interview should be regarded as matters of opinion and not as representations of fact. We are unable to guarantee with certainty the veracity of any of the statements made by the speakers in this video. This video should be regarded as an honest review of our company’s products and services from the perspective of a real user of our company’s products and services. This video was prepared for general informational purposes only and our company assumes no liability for any errors or omissions in the content, or for any actions taken based on the information provided. The speakers in this video received no compensation, reward or any other form of inducement from our company to participate in this interview or to endorse any of our products or services. This video and any content contained within this video is protected by copyright law. Unauthorized use or distribution is strictly prohibited.
2026.02.20 10:43
-
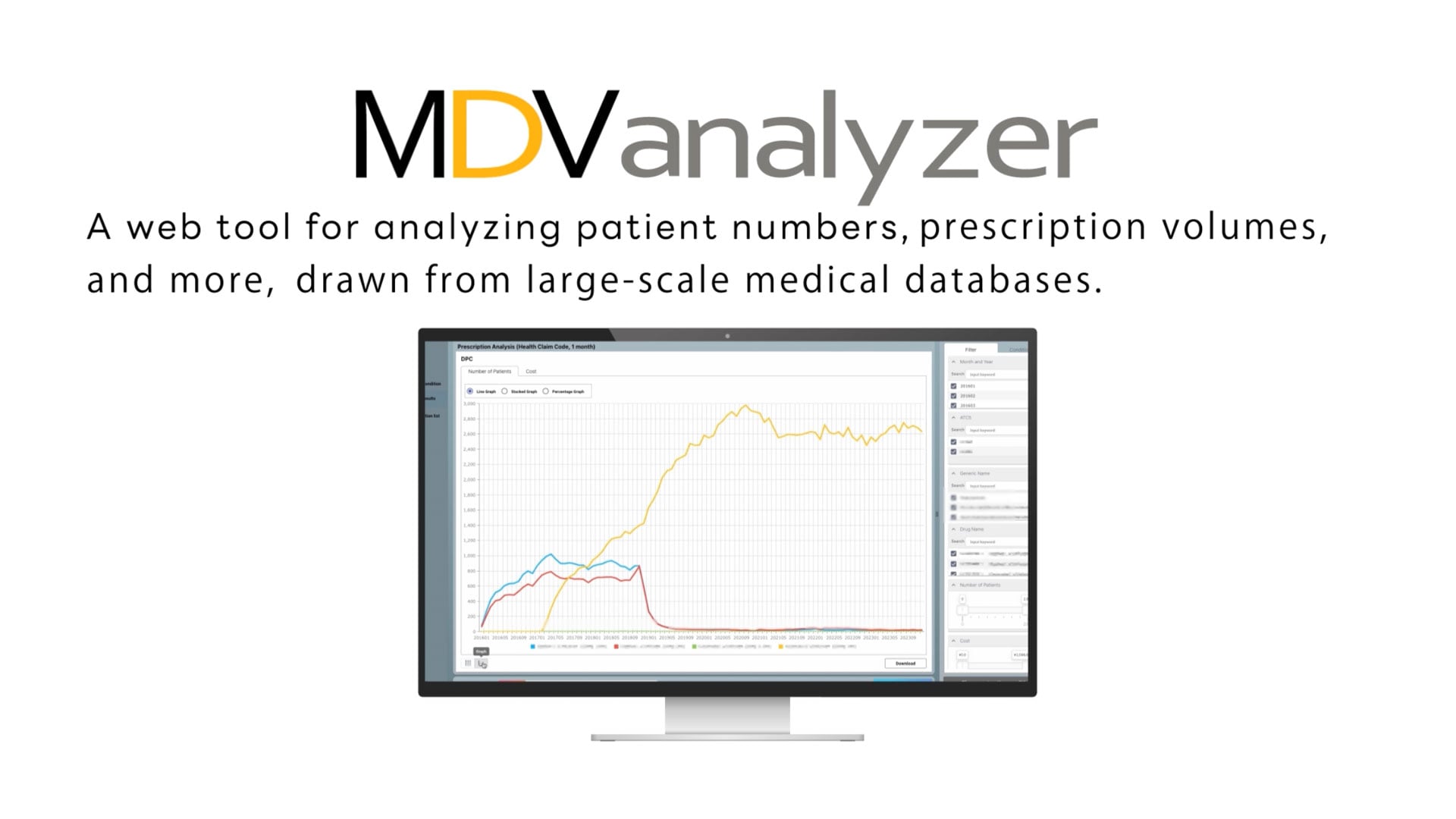
【MDV analyzer】 a web tool featuring analyses for patient count, treatment volume, and more with a medical database
Service details page>> https://en.mdv.co.jp/ebm/service/mdvanalyzer/ MDV analyzer, a web tool featuring analyses for patient count, treatment volume, and more with a medical database Analyses for patient count, prescription duration, treatment volume, and more based on Japan’s largest medical database are available. In addition to diseases and medications, the database also supports multifaceted analyses based on actual medical procedures, such as surgeries and tests. Feature Overview All-round usage from development to regulatory submission, not to mention marketing strategy and understanding the target population for database study. Standardized analyses are included to address time consuming procedures from definition to delivery that occurs with ad-hoc service. *Data can be disclosed to a third party upon a disclosure request.
2025.10.01 05:43
Databases, data analysis requests, and more.
© Medical Data Vision Co., Ltd. All Rights Reserved.




